Some of Our Projects
Cloud-Connected Controller for Insulin Pump

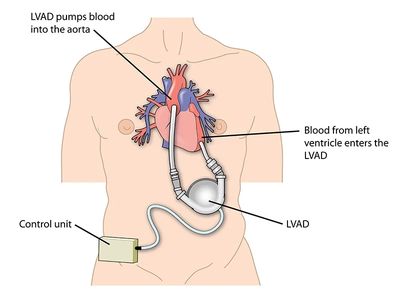
Safety Architecture for Life-Critical Active Implant

Translation Acceleration Platform & Automated GUI Validation
Translation Acceleration Platform & Automated GUI Validation

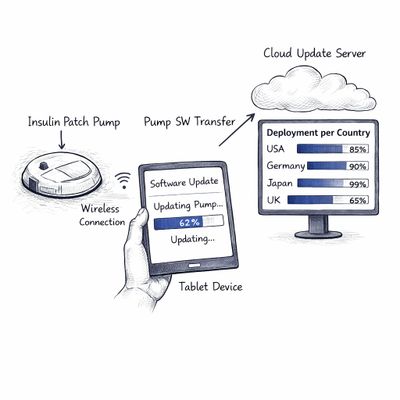
Cloud-Connected Controller for Insulin Pump
Co-development · SiMD · Cloud Architecture · FDA · CE Mark
An insulin pump manufacturer had developed breakthrough core delivery technology, but the pump itself lacked the connectivity and intelligence to unlock its full clinical potential. 4ms joined as a co-development partner, contributing system architecture and end-to-end software engineering to build a cloud-connected controller that served as the bridge between the physical device and the digital ecosystem.
The result went well beyond connectivity. The platform we built enabled advanced therapy configurations impossible on-device, real-world usage analytics for clinicians and product teams, and over-the-air software updates - turning a standalone device into an intelligent, continuously improving system. Delivered as a turnkey project, the delivery system achieved both FDA clearance and CE marking.
4ms contribution: Full Sw life cycle development, System architecture, cloud bridge design, full ownership from concept to registration.
Safety Architecture for Life-Critical Active Implant
Risk Management · DFMEA · Design Mitigations · Clinical Studies
Our client had developed an active implantable device targeting a critical cardiac condition — a high-stakes program where safety architecture directly determines patient outcomes. 4ms was brought in at the risk management phase, leading a rigorous DFMEA process that mapped failure modes across the full system.
What distinguished this engagement was what happened next: the DFMEA didn't just document risk — it drove design. Identified failure modes led to concrete mitigations, with 4ms designing and implementing a portion of those mitigations directly, others developed by the client, and others built jointly. This deeply embedded co-development model ensured safety was engineered into the architecture, not added as a layer on top. The device is now in clinical studies.
4ms contribution:Risk assessment leadership, DFMEA facilitation, design mitigation development — embedded at the intersection of safety engineering and product design.
Translation Acceleration Platform & Automated GUI Validation
SW Tools · Localization Engineering · Test Automation · Time-to-Market
Entering new markets requires localized products, but for regulated medical device software, the traditional localization workflow is a significant liability. Each language cycle demands translation, developer integration, QA testing, and back-and-forth review. Multiplied across five, ten, or fifteen languages, this becomes a major program-level bottleneck that delays market entry and strains engineering resources.
4ms identified the root cause: translators were working blind, with no visibility into how their text would actually render on the device. We designed and built a purpose-built translation aiding tool that renders the real product GUI live as text is entered, so translators catch layout breaks, truncations, and overlaps immediately, in context, without a single development cycle.
As a second phase, we extended the platform with an automated testing system that compares every language file against the GUI and executes a full configurable test suite - systematically validating every string, in every language, against every relevant use case.
4ms contribution: Full-stack tool design and development from workflow analysis through production-ready localization and automated validation tooling. Measurable reduction in localization cycle time and engineering overhead.

This website uses cookies.
We use cookies to analyze website traffic and optimize your website experience. By accepting our use of cookies, your data will be aggregated with all other user data.